EP Edge Newsletter Part II: Pulsed Field Ablation Trials — ADVENT, PULSED AF, Sphere-9, VARIPULSE, VOLT, INSIGHT, and PULSAR
カートのアイテムが多すぎます
カートに追加できませんでした。
ウィッシュリストに追加できませんでした。
ほしい物リストの削除に失敗しました。
ポッドキャストのフォローに失敗しました
ポッドキャストのフォロー解除に失敗しました
-
ナレーター:
-
著者:
概要
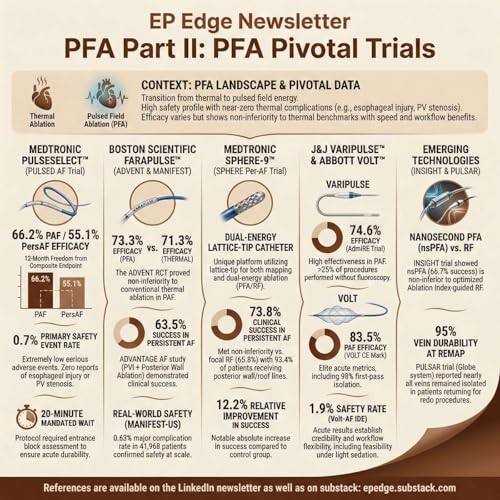
In this episode of the EP Edge Newsletter Podcast, we analyze the pivotal clinical trials that established pulsed field ablation (PFA) as a major technological advancement in atrial fibrillation ablation. We examine the foundational FDA-approval studies, including the PULSED AF trial evaluating the Medtronic PulseSelect system and the ADVENT randomized trial comparing FARAPULSE pulsed field ablation with conventional radiofrequency and cryoballoon ablation. These trials demonstrated that pulsed field ablation achieves high rates of acute pulmonary vein isolation with freedom from atrial arrhythmia recurrence comparable to thermal ablation, while maintaining a favorable safety profile and procedural efficiency. Importantly, these studies defined modern clinical endpoints for catheter ablation success, including composite measures incorporating arrhythmia recurrence, antiarrhythmic drug use, cardioversion, and repeat ablation.
PFA Part II
We also explore next-generation pulsed field ablation platforms, including the Sphere-9 lattice-tip catheter, VARIPULSE system, and VOLT balloon-in-basket catheter, each designed to improve lesion durability, catheter stability, and procedural reproducibility. These studies demonstrate consistently high rates of pulmonary vein isolation, favorable safety outcomes, and evolving workflow advantages, including reduced fluoroscopy use and efficient lesion delivery. We then examine the latest randomized and investigational trials, including INSIGHT, which compared nanosecond pulsed field ablation with optimized ablation-index guided radiofrequency ablation, and the PULSAR IDE trial evaluating spherical array catheter design for durable pulmonary vein isolation. These trials highlight a critical reality in atrial fibrillation ablation: long-term success is determined not only by energy source, but by catheter design, tissue contact, lesion geometry, and pulmonary vein isolation durability.
PFA Part II
Full references, detailed discussion, figures, and graphical summaries are available on the EP Edge Newsletter on LinkedIn, as well as the complete long-form article on Substack at epedge.substack.com. If you have questions, suggestions, or feedback, please contact epedgecast@gmail.com.


