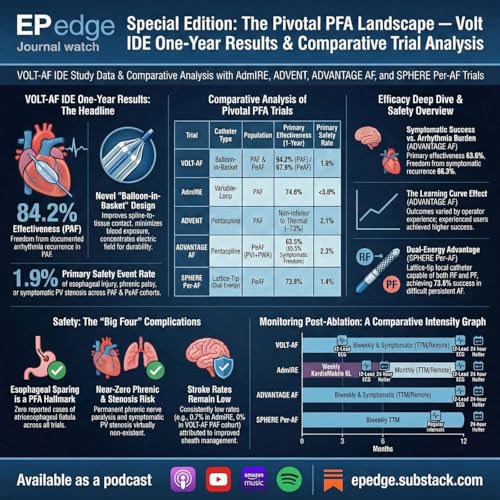
EP Edge Journal Watch Special Edition: VOLT IDE One-Year Results in Context: ADVENT, ADVANTAGE-AF, AdmIRE, SPHERE-Per-AF & U.S. PFA Pivotal Trials
カートのアイテムが多すぎます
カートに追加できませんでした。
ウィッシュリストに追加できませんでした。
ほしい物リストの削除に失敗しました。
ポッドキャストのフォローに失敗しました
ポッドキャストのフォロー解除に失敗しました
-
ナレーター:
-
著者:
概要
Pulsed field ablation (PFA) has rapidly reshaped atrial fibrillation ablation, but true clinical validation depends on durable one-year outcomes, not early feasibility or acute safety alone. In this EP Edge Journal Watch Special Edition, Dr. Niraj Sharma delivers a comprehensive, system-level analysis of the Abbott VOLT IDE one-year data, now completing the 12-month efficacy and safety landscape across major U.S. PFA platforms.
This episode critically reviews and contextualizes results from the VOLT IDE trial alongside other pivotal and near-pivotal studies, including ADVENT, ADVANTAGE-AF, AdmIRE, SPHERE-9 first-in-human, and SPHERE-Per-AF. The discussion spans paroxysmal, persistent, and advanced AF populations, with attention to how ablation strategy (PVI-only vs adjunctive lesions), patient risk profile, and post-ablation monitoring intensity influence reported outcomes.
Key themes include one-year efficacy versus composite effectiveness, freedom from atrial arrhythmias, repeat procedures, and major safety endpoints such as stroke, tamponade, and esophageal injury. Rather than ranking technologies, this episode emphasizes methodology, trial design, and clinical context, explaining why efficacy signals differ across studies and how these data should be interpreted in everyday electrophysiology practice.
For additional references, detailed tables, graphics, and deeper comparative analysis, visit the LinkedIn EP Edge newsletter and Substack at ephedge.substack.com. If you have suggestions or concerns, you can reach Dr. Sharma at ephedgecast@gmail.com


